We are expanding
our material know-how
and test services with
the DEKRA laboratories!
Find out more in our
press release.
News:
Titanium alloys and titanium purity of medical instruments and implants according to
Accredited test laboratory
for materials testing, product testing & failure analysis.
Your specialist for metals,
plastics, elastomers.
We are expanding
our material know-how
and test services with
the DEKRA laboratories!
Find out more in our
press release.
News:
Titanium alloys and titanium purity of medical instruments and implants
according to DIN EN ISO 832
Accredited test laboratory
to DIN EN ISO / IEC 17025.
Your specialist for metals,
plastics, elastomers.
DSC analysis - differential scanning calorimetry.
In accordance with the multitude of possible uses of the DSC analysis we advise you comprehensively on the optimal solution for your situation. We clarify the analytical tasks with you, perform the analyzes, and provide a professional interpretation of the results within a detailed report.
Do you know the melting behavior or the crystallinity of your plastic parts? Are foreign material and admixtures in plastics an issue for you?
We can give you the answers to your questions through DSC analysis tests:
Do you know the melting behavior or the crystallinity of your plastic parts? Are foreign material and admixtures in plastics an issue for you?
We have the answers to your questions through investigations with DSC analysis:
- Plastic provisions
- Melting behavior / crystallization behavior
- Solid-state reactions
- Glass transitions
- Polymorphic Conversions
- Degree of crystallinity
- Crosslinking reactions
- Oxidation stability
- Decomposition behavior
- Determination of purity

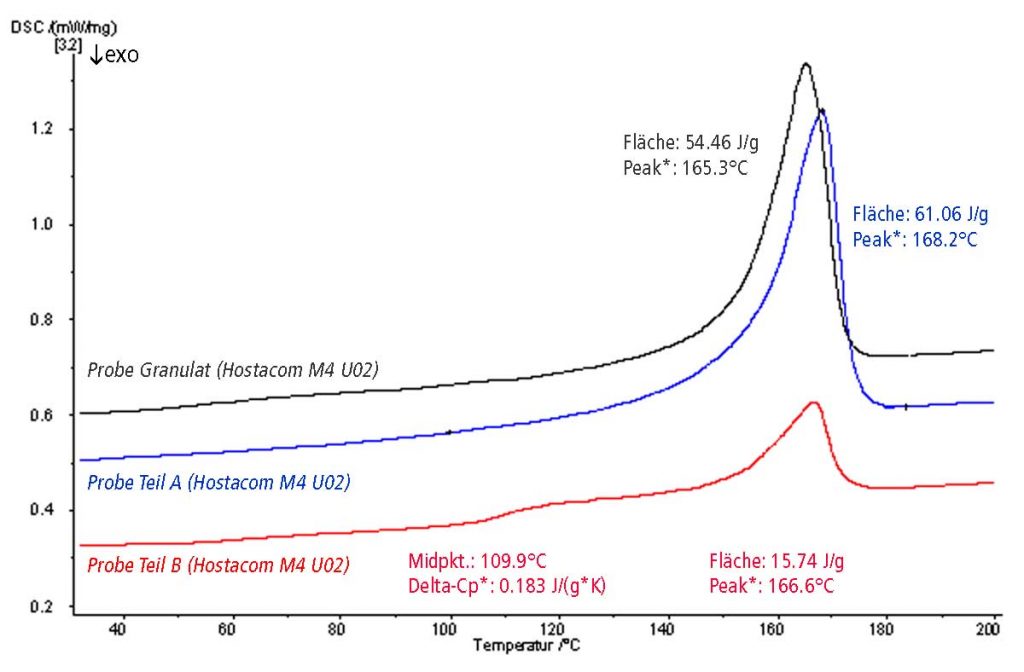
The graphic shows the first heating curves of 3 samples in direct comparison.
In addition to the melting effect, sample part 3 also has a glass transition, which suggests the presence of polystyrene, for example. So this part was not made from pure Hostacom M4 U02. This finding is confirmed by the second heating curve.
Sample part B clearly shows a glass transition for an amorphous polymer component.
The DSC results for part A almost correspond to the results for the granules, so part A was definitely made from Hostacom M4 U02.
In contrast, part B had a different starting material (mixed with PS as a foreign material).